Advertisements
Advertisements
प्रश्न
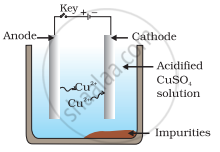
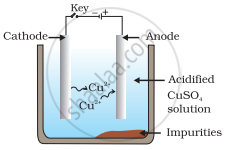
Which one of the following figures correctly describes the process of electrolytic refining?
विकल्प
उत्तर

In the process of electrolytic refining of copper, the impure copper metal is made the anode and connected to positive terminal of the battery. A thin strip of pure copper metal is made the cathode and connected to negative terminal of the battery. A solution of the metal salt (CuSO4) is used as an electrolyte. On passing the current through the electrolyte, the pure metal from the anode dissolves into the electrolyte as Cu2+. An equivalent amount of pure metal from the electrolyte is deposited on the cathode. The soluble impurities go into the solution, whereas, the insoluble impurities settle down at the bottom of the anode and are known as anode mud.
APPEARS IN
संबंधित प्रश्न
What is meant by refining of a metal? Name the most widely used method for the refining of impure metals obtained by various reduction processes. Describe this method with the help of a labelled diagram by taking the example of any metal.
Explain how the following metals are obtained from their compounds by the reduction process:
(i) Metal M which is in the middle of the reactivity series.
(ii) Metal N which is high up in the reactivity series.
Give one example of each type
Draw a neat labelled diagram.
Magnetic separation method.
Draw a neat labelled diagram.
Hydraulic separation method.
Name one metal each occurring as :
A sulphine
The lightest liquid metal is ____________.
During electrolytic refining of zinc, it gets
Identify the terms for the following:
The electrode where reduction takes place.
A reddish-brown metal ‘X’, when heated in air, gives a black compound ‘Y’, which when heated in the presence of H2 gas gives ‘X’ back. ‘X’ is refined by the process of electrolysis; this refined form of ‘X’ is used in electrical wiring. Identify ‘X’ and ‘Y’. Draw a well-labelled diagram to represent the process of refining ‘X’.
Answer the following question with reference to the electrorefining of copper:
What is the anode made of?