Advertisements
Advertisements
प्रश्न
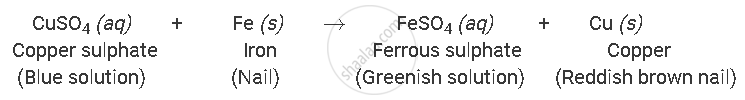
Why does the colour of copper sulphate solution change when an iron nail is kept immersed in it?
उत्तर
The colour of copper sulphate solution changes when iron nail is kept immersed in it because displacement reaction occurs between copper sulphate and iron to produce ferrous sulphate solution, which is green in colour.
APPEARS IN
संबंधित प्रश्न
\[\ce{Fe2O3 + 2Al -> Al2O3 + 2Fe}\]
The above reaction is an example of a ______.
what type of reaction is the following:
Cl2 + 2KI → 2KCl + I2
Explain the following type of chemical reaction, giving two examples for it:
Displacement reaction
Name the type of reaction which is governed by the position of a metal in the metal activity series ?
Give reasons for the following:
The reaction of iron (III) oxide [Fe2O3] with heated aluminum is used to join cracked machine parts.
Which of the following is a displacement reaction?
\[\ce{Pb + CuCl2 -> PbCl2 + Cu}\]
The above reaction is an example of:
Complete the given chemical equation:
\[\ce{Zn(s) + CuSO4(aq) -> \underline{}\underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}\underline{}}\]
Name the type of reaction.
State the change in colour observed in following case mentioning the reason:
A piece of zinc is dipped in ferrous sulphate solution.
The colour of the solution observed after 30 minutes of placing zinc metal in copper sulphate solution is ______.
