Advertisements
Advertisements
प्रश्न
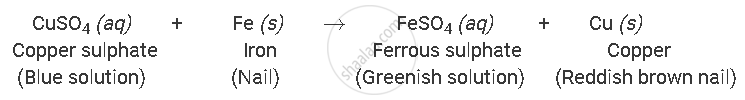
Why does the colour of copper sulphate solution change when an iron nail is kept immersed in it?
उत्तर
The colour of copper sulphate solution changes when iron nail is kept immersed in it because displacement reaction occurs between copper sulphate and iron to produce ferrous sulphate solution, which is green in colour.
APPEARS IN
संबंधित प्रश्न
Reaction of iron nails with copper sulphate solution is an example of
(a) Combination reaction
(b) Decomposition reaction
(c) Displacement reaction
(d) Double displacement reaction
State two characteristics of the chemical reaction which takes place when dilute sulphuric acid is poured over zinc granules.
What happens when a piece of iron metal is placed in copper sulphate solution? Name the type of reaction involved.
When a black metal compound XO is heated with a colourless gas Y2, then metal X and another compound Y2O are formed. Metal X is red-brown in colour which does not react with dilute acids at all. Gas Y2 can be prepared by the action of a dilute acid on any active metal. The compound Y2O is a liquid at room temperature which can turn anhydrous copper sulphate blue.
(a) What do you think is metal X?
(b) What could be gas Y2?
(c) What is compound XO?
(d) What is compound Y2O?
(e) Write the chemical equation of the reaction which takes place on heating XO with Y2.
(f) What type of chemical reaction is illustrated in the above equation?
Explain the following type of chemical reaction, giving two examples for it:
Displacement reaction
i) Observe the following reaction and answer the following questions.
`CuSO_4 (aq) + Fe (s) → FeSO_4(aq) + Cu (s)`
a) Identify and write the type of chemical reaction.
b) Write the definition of above reaction.
Classify the following reaction into different type:
Zn + CuSO4 → ZnSO4 + Cu
Select the correct answer for the statement given below:
The displaced product of the displacement reaction of sodium with cold water.
Match the following
Identify the types of reactions.
| REACTION | TYPE |
| NH4OH(aq) + CH3COOH(aq) → CH3COONH4(aq) + H2O(l) | Single Displacement |
| Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s) | Combustion |
| \[\ce{ZnCO3_{(s)} + ->[Heat]ZnO_{(s)} + CO2_{(g)}}\] | Neutralization |
| C2H4(g) + 4O2(g) → 2CO2(g) + 2H2 O(g) + Heat | Thermal decomposition |
State the change in colour observed in following case mentioning the reason:
A piece of zinc is dipped in ferrous sulphate solution.
