Advertisements
Advertisements
प्रश्न
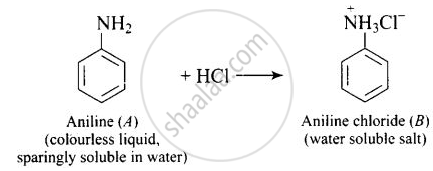
Why is aniline soluble in aqueous HCl?
उत्तर
Aniline forms anilinium chloride salt with aqueous HCl which is soluble in water.
APPEARS IN
संबंधित प्रश्न
Why does acetylation of –NH2 group of aniline reduce its activating effect?
Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
A colourless substance ‘A’ \[\ce{(C6H7N)}\] is sparingly soluble in water and gives a water soluble compound ‘B’ on treating with mineral acid. On reacting with \[\ce{CHCl3}\] and alcoholic potash ‘A’ produces an obnoxious smell due to the formation of compound ‘C’. Reaction of ‘A’ with benzenesulphonyl chloride gives compound ‘D’ which is soluble in alkali. With \[\ce{NaNO2}\] and \[\ce{HCl}\], ‘A’ forms compound ‘E’ which reacts with phenol in alkaline medium to give an orange dye ‘F’. Identify compounds ‘A’ to ‘F’.
Which of the following is the most stable diazonium salt?
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
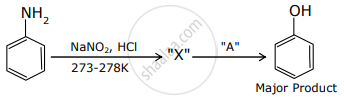
Major Product In the above chemical reaction, intermediate "X" and reagent/condition "A" are:
Aniline dissolved in dilute HCl is reacted with sodium nitrate at 0 °C. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is:
How will the following be converted? (Give chemical equation)
Aniline to benzene diazonium chloride.
