Advertisements
Advertisements
प्रश्न
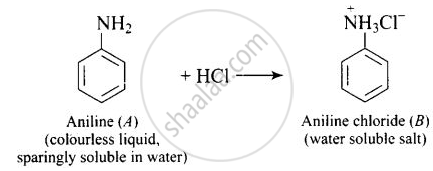
Why is aniline soluble in aqueous HCl?
उत्तर
Aniline forms anilinium chloride salt with aqueous HCl which is soluble in water.
APPEARS IN
संबंधित प्रश्न
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Why does acetylation of –NH2 group of aniline reduce its activating effect?
Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
A colourless substance ‘A’ \[\ce{(C6H7N)}\] is sparingly soluble in water and gives a water soluble compound ‘B’ on treating with mineral acid. On reacting with \[\ce{CHCl3}\] and alcoholic potash ‘A’ produces an obnoxious smell due to the formation of compound ‘C’. Reaction of ‘A’ with benzenesulphonyl chloride gives compound ‘D’ which is soluble in alkali. With \[\ce{NaNO2}\] and \[\ce{HCl}\], ‘A’ forms compound ‘E’ which reacts with phenol in alkaline medium to give an orange dye ‘F’. Identify compounds ‘A’ to ‘F’.
Predict the reagent or the product in the following reaction sequence.
In order to distinguish between C2H5NHz and C6H5NHz, which of the following reagents is useful?
Which of the following is true characteristic feature of aniline?
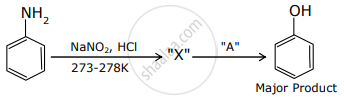
Major Product In the above chemical reaction, intermediate "X" and reagent/condition "A" are:
Aniline dissolved in dilute HCl is reacted with sodium nitrate at 0 °C. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is:
Identify A and B for the following reaction:

What are polymers?
