Advertisements
Advertisements
प्रश्न
Write an equation for the laboratory preparation of an unsaturated hydrocarbon from calcium carbide.
उत्तर
\[\ce{\underset{Calcium carbide}{CaC2} + \underset{Water}{2H2O} -> \underset{Calcium hydroxide}{Ca(OH)2} + \underset{Acetylene }{C2H2}\uparrow}\]
APPEARS IN
संबंधित प्रश्न
Choose the correct answer from the options given below:
If the molecular formula of an organic compound is C10H18 it is:
1) alkene
2) alkane
3) alkyne
4) Not a hydrocarbon
The compound formed where two alkyl groups are linked by 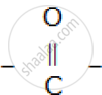 group.
group.
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulphide, ethyne]
This gas is used for welding purposes.
State the conditions required for the given reaction to take place:
Preparation of ethyne from ethylene dibromide
Write the structure of the following compound:
2 – methylpropane
Name the products formed and write an equation when ethyne is added to the bromine in an
inert solvent ?
Fill in the blank with appropriate word/words.
The general formula for alkene and alkyne are _____ and______respectively
Draw the structural formula for the following:
But-2-yne
Name the hydrocarbon which will give acetylene (ethyne) gas when treated with water.
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
hydrogen
