Advertisements
Advertisements
प्रश्न
Write the basic features of photon picture of electromagnetic radiation on which Einstein’s photoelectric equation is based.
उत्तर
Photon picture of electromagnetic radiation on which Einstein’s photoelectric equation is based on particle nature of light. Its basic features are:
(i) In interaction with matter, radiation behaves as if it is made up of particles called photons.
(ii) Each photon has energy E (=hν) and momentum p (=hν/c), and speed c, the speed of light.
(iii) All photons of light of a particular frequency ν, or wavelength λ, have the same energy E (=hν=hc/λ) and momentum p (=hν/c=h/λ), whatever the intensity of radiation may be.
(iv) By increasing the intensity of light of given wavelength, there is only an increase in the number of photons per second crossing a given area, with each photon having the same energy. Thus, photon energy is independent of intensity of radiation.
(v) Photons are electrically neutral and are not deflected by electric and magnetic fields.
(vi) In a photon-particle collision (such as photon-electron collision), the total energy and total momentum are conserved. However, number of photons may not be conserved.
APPEARS IN
संबंधित प्रश्न
When radiations of wavelength λ1 and λ2 are incident on certain photosensitive, such that E1 > E2 . Then Planck's constant 'h' is ......................... .
(C = Velocity of light).
Find the value of energy of electron in eV in the third Bohr orbit of hydrogen atom.
(Rydberg's constant (R) = 1· 097 x 107m - 1,Planck's constant (h) =6·63x10-34 J-s,Velocity of light in air (c) = 3 x 108m/ s.)
With reference to the photoelectric effect, define threshold wavelength
According to Einstein’s model, the threshold frequency for a metal having work function ϕ0 is given by _________.
Plot a labelled graph of |Vs| where Vs is stopping potential versus frequency f of the incident radiation. State how will you use this graph to determine the value of Planck's constant?
In an inelastic collision, which of the following does not remain conserved?
Dimensions of ‘resistance’ are same as (where h is Planck's constant and e is charge):
Which of the following is/are true for cathode ray
- Calculate the frequency of a photon of energy 6.5 × 10−19 J.
- Can this photon cause the emission of an electron from the surface of Cs of work function 2.14 eV? If yes, what will be the maximum kinetic energy of the photoelectron?
The graphs below show the variation of the stopping potential VS with the frequency (ν) of the incident radiations for two different photosensitive materials M1 and M2.
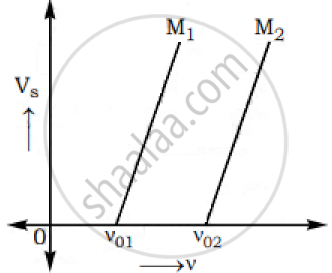
Express work function for M1 and M2 in terms of Planck’s constant(h) and Threshold frequency and charge of the electron (e).
If the values of stopping potential for M1 and M2 are V1 and V2 respectively then show that the slope of the lines equals to `(V_1-V_2)/(V_(01)-V_(02))` for a frequency,
ν > ν02 and also ν > ν01
