Advertisements
Advertisements
प्रश्न
Using Bohr’s postulates, obtain the expressions for (i) kinetic energy and (ii) potential energy of the electron in stationary state of hydrogen atom.
Draw the energy level diagram showing how the transitions between energy levels result in the appearance of Lymann Series.
उत्तर
According to Bohr’s postulates, in a hydrogen atom, a single alectron revolves around a nucleus of charge +e. For an electron moving with a uniform speed in a circular orbit os a given radius, the centripetal force is provided by Columb force of attraction between the electron and the nucleus. The gravitational attraction may be neglected as the mass of electron and proton is very small.
So,
`mv^2/r = (ke^2)/r^2`
0r `mv^2 = (ke^2)/r ...................(1)`
where m = mass of electron
r = radius of electronic orbit
v = velocity of electron.
Again,
`mvr = (nh)/(2π)`
` or v = (nh)/(2πmr)`
From eq(1), we get,
`m ((nh)/(2πmr))^2 = (ke^2)/r`
`⇒ r = (n^2h^2)/(4π^2kme^2) .....................(2) `
(i) Kinetic energy of electron,
`E_k = 1/2mv^2 = (ke^2)/(2r)`
Using eq (2), we get
`E_k = (ke^2)/2 (4π^2kme^2)/(n^2h^2)`
= `(2π^2k^2me^4)/(n^2h^2)`
(ii) Potential energy
`E^p= - ke^2 xx (4π^2k^2me^4)/(n^2h^2) `
Energy level diagram showing the transitions between energy levels result in the appearance ofLymann series:
For Lymann series, nf = 1 and ni = 2, 3, 4, 5, …
`1/λ = R_H(1/I^2 - 1/n^2)`
Where, ni = 2, 3, 4, 5, …
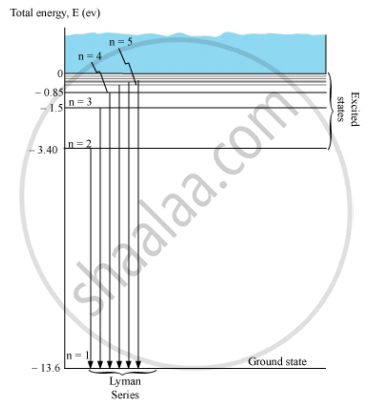
APPEARS IN
संबंधित प्रश्न
Draw a neat, labelled energy level diagram for H atom showing the transitions. Explain the series of spectral lines for H atom, whose fixed inner orbit numbers are 3 and 4 respectively.
The longest wavelength doublet absorption transition is observed at 589 and 589.6 nm. Calculate the frequency of each transition and energy difference between two excited states.
A neutron moving with a speed υ strikes a hydrogen atom in ground state moving towards it with the same speed. Find the minimum speed of the neutron for which inelastic (completely or partially) collision may take place. The mass of neutron = mass of hydrogen = 1.67 × 10−27 kg.v
The light emitted in the transition n = 3 to n = 2 in hydrogen is called Hα light. Find the maximum work function a metal can have so that Hα light can emit photoelectrons from it.
The spectral line obtained when an electron jumps from n = 5 to n = 2 level in hydrogen atom belongs to the ____________ series.
Which of the following is/are CORRECT according to Bohr's atomic theory?
(I) Energy is emitted when electron moves from a higher stationary state to a lower one.
(II) Orbits are arranged concentrically around the nucleus in an increasing order of energy.
(III) The energy of an electron in the orbit changes with time.
Consider two different hydrogen atoms. The electron in each atom is in an excited state. Is it possible for the electrons to have different energies but same orbital angular momentum according to the Bohr model? Justify your answer.
Use Bohr's postulate to prove that the radius of nth orbit in a hydrogen atom is proportional to n2.
What is meant by ionisation energy?
The figure below is the Energy level diagram for the Hydrogen atom. Study the transitions shown and answer the following question:
- State the type of spectrum obtained.
- Name the series of spectrum obtained.

