Advertisements
Advertisements
प्रश्न
Using Bohr’s postulates, obtain the expressions for (i) kinetic energy and (ii) potential energy of the electron in stationary state of hydrogen atom.
Draw the energy level diagram showing how the transitions between energy levels result in the appearance of Lymann Series.
उत्तर
According to Bohr’s postulates, in a hydrogen atom, a single alectron revolves around a nucleus of charge +e. For an electron moving with a uniform speed in a circular orbit os a given radius, the centripetal force is provided by Columb force of attraction between the electron and the nucleus. The gravitational attraction may be neglected as the mass of electron and proton is very small.
So,
`mv^2/r = (ke^2)/r^2`
0r `mv^2 = (ke^2)/r ...................(1)`
where m = mass of electron
r = radius of electronic orbit
v = velocity of electron.
Again,
`mvr = (nh)/(2π)`
` or v = (nh)/(2πmr)`
From eq(1), we get,
`m ((nh)/(2πmr))^2 = (ke^2)/r`
`⇒ r = (n^2h^2)/(4π^2kme^2) .....................(2) `
(i) Kinetic energy of electron,
`E_k = 1/2mv^2 = (ke^2)/(2r)`
Using eq (2), we get
`E_k = (ke^2)/2 (4π^2kme^2)/(n^2h^2)`
= `(2π^2k^2me^4)/(n^2h^2)`
(ii) Potential energy
`E^p= - ke^2 xx (4π^2k^2me^4)/(n^2h^2) `
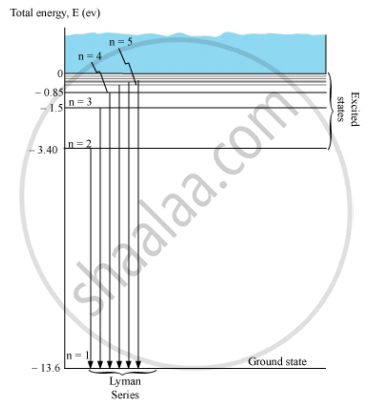
Energy level diagram showing the transitions between energy levels result in the appearance ofLymann series:
For Lymann series, nf = 1 and ni = 2, 3, 4, 5, …
`1/λ = R_H(1/I^2 - 1/n^2)`
Where, ni = 2, 3, 4, 5, …
APPEARS IN
संबंधित प्रश्न
If the velocity of the electron in Bohr’s first orbit is 2.19 × 106 ms-1, calculate the de Broglie wavelength associated with it.
Using Bohr’s postulates for hydrogen atom, show that the total energy (E) of the electron in the stationary states tan be expressed as the sum of kinetic energy (K) and potential energy (U), where K = −2U. Hence deduce the expression for the total energy in the nth energy level of hydrogen atom.
Find the wavelength of the radiation emitted by hydrogen in the transitions (a) n = 3 to n= 2, (b) n = 5 to n = 4 and (c) n = 10 to n = 9.
Radiation coming from transition n = 2 to n = 1 of hydrogen atoms falls on helium ions in n = 1 and n = 2 states. What are the possible transitions of helium ions as they absorbs energy from the radiation?
If l3 and l2 represent angular momenta of an orbiting electron in III and II Bohr orbits respectively, then l3: l2 is :
Draw energy level diagram for a hydrogen atom, showing the first four energy levels corresponding to n=1, 2, 3 and 4. Show transitions responsible for:
(i) Absorption spectrum of Lyman series.
(ii) The emission spectrum of the Balmer series.
The mass of a H-atom is less than the sum of the masses of a proton and electron. Why is this?
Use Bohr's postulate to prove that the radius of nth orbit in a hydrogen atom is proportional to n2.
What is the energy of an electron in stationary state corresponding to n = 2?
The de Broglie wavelength of an electron in the first Bohr’s orbit of hydrogen atom is equal to ______.
