Advertisements
Advertisements
प्रश्न
Write a chemical reaction to show the dehydration of ethanol.
उत्तर
Ethanol undergoes dehydration to form ethane.
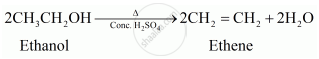
APPEARS IN
संबंधित प्रश्न
How can ethanol and ethanoic acid be differentiated on the basis of their physical properties?
Write balanced chemical equations for Preparation of ethanol from Ethyl Chloride
State two used of ethanol (other than as a fuel).
Addition reactions and substitution reactions are types of organic reactions.Which type of reaction is shown by?
Ethene?
Write a balanced chemical equation for the following:
Ethanol reacts with sodium at room temperature
Write the uses of ethanol.
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the reactions.
Neutralization of NaOH with ethanoic acid.
