Advertisements
Advertisements
Question
Write a chemical reaction to show the dehydration of ethanol.
Solution
Ethanol undergoes dehydration to form ethane.
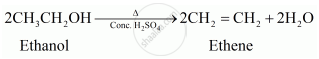
APPEARS IN
RELATED QUESTIONS
A carbon compound 'P' on heating with excess conc. H2SO4 forms another carbon compound 'Q' which on addition of hydrogen in the presence of nickel catalyst forms a saturated carbon compound 'R'. One molecule of 'R' on combustion forms two molecules of carbon dioxide and three molecules of water. Identify P, Q and R and write chemical equations for the reactions involved
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
How is ethanol prepared by fermentation?
Ethanol can be converted into ethene which can be changed into ethane. Choose the correct word or phrase from the bracket to complete the following sentence.
The conversion of ethanol into ethene is an example of ______. (dehydration, dehydrogenation).
In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO3 test
(iv) Ester test
The organic compound obtained as the end product of the fermentation of a sugar solution is ______.
Complete the following activity.
| Boiling point of ethanol | → | |
| General name of ethanol | → | |
| Use of ethanol | → | |
| Boiling point of ethanoic acid | → | |
| Melting point of pure ethanoic acid | → |
Which of the following are used as anesthetics?
How is ethanol manufactured from sugarcane?
______ react with dil. HNO3 to evolve hydrogen gas.
