Advertisements
Advertisements
प्रश्न
Write structure of adipic acid.
उत्तर
\[\begin{array}{cc}
\phantom{}\ce{O}\phantom{...............................}\ce{O}\phantom{}\\
\phantom{}||\phantom{................................}||\phantom{}\\
\phantom{}\ce{\underset{Hexanedioic acid}{HO - C - CH2} - CH2 - CH2 - \underset{(Adipic acid)}{CH2 - C - OH}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Formic acid is stronger than acetic acid. Explain.
Write the reducing agent which CANNOT reduce –COOH group.
What is the action of following on proponal?
Hydrazine
Write the preparation reactions for acid amide from the following.
Carboxylic acid
Write the preparation reactions for acid amide from the following.
Acid chloride
Write reactions for the following conversions.
4-Nitrobenzoic acid to Nitrobenzene
Write reaction for preparation of acetophenone from benzoyl chloride.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Ethane-1,2-diol in presence of dry HCl.
Complete the following sequence of reactions and write structures for A, B, C.
\[\ce{Dry ice ->[i. Dry ether][ii. Hydrolysis] A ->[PCl5] B ->[H2(gas)][Pd - BaSO4] C}\]
In the following reaction:
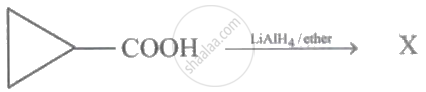
The product X is:
A flavouring agent found in oil of wintergreen is ______.
Which of the following carboxylic acids will have the highest acidity?
What amount of dinitrogen contains 3.6 × 1018 molecules?
The reducing agent preferred to convert carboxylic acids and esters to primary alcohols is ____________.
Which among the following is the strongest acid?
The oxidation number of B in NaBH4 is ____________.
Which of the following functional groups is reduced by diborane?
Which of the following compounds is obtained when ethanoic anhydride is treated with water?
Which of the following carboxylic acids ha highest acidic strength?
The number of bonding pairs and lone pairs of electrons present in the central atom of ammonia molecular are respectively.
Which of the following can reduce?
\[\ce{RCOOH -> RCH2OH}\]
Draw structures 01 conjugate bases of monochloroacetic acid and dichloroacetic acid. Which one is more stabilized by -I effect?
How sodium bicarbonate test is used to distinguish between carboxylic acid and phenol?
How will you prepare Acetic anhydride from acetic acid?
