Advertisements
Advertisements
प्रश्न
Write structures of various carbocations that can be obtained from 2-methylbutane. Arrange these carbocations in order of increasing stability.
उत्तर
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - \overset{+}{C}H2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{......} \end{array}\] |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH - \overset{+}{C}H - CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{......} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3 - \overset{+}{C} - CH2 - CH3}\\ |\phantom{.......}\\ \ce{CH3}\phantom{......} \end{array}\] |
| (iv) | \[\begin{array}{cc} \ce{\overset{+}{C}H2 - CH - CH2 - CH3}\\ |\phantom{.......}\\ \ce{CH3}\phantom{......} \end{array}\] |
The stability order is found to be: (III) > (II) > (I) > (IV)
This is because (III) is tertiary carbocation, (II) is secondary carbocation, (I) and (IV) are primary.
APPEARS IN
संबंधित प्रश्न
For the following bond cleavages, use curved arrows to show the electron flow and classify homolysis or heterolysis. Identify reactive intermediate produced as free radical, carbocation and carbanion.
\[\ce{CH3O - OCH3 -> CH3\overset\bullet{\text{O}} + \overset\bullet{\text{O}}CH3}\]
For the following bond cleavages, use curved-arrows to show the electron flow and classify as homolysis or heterolysis. Identify reactive intermediate produced as free radical, carbocation and carbanion.
For the following bond cleavages, use curved-arrows to show the electron flow and classify as homolysis or heterolysis. Identify reactive intermediate produced as free radical, carbocation and carbanion.
Which of the following carbocation is most stable?
What is the correct order of decreasing stability of the following cations.
| I. | \[\ce{CH3 - \overset{⊕}{C}H - CH3}\] |
| II. | \[\ce{CH3 - \overset{⊕}{C}H - OCH3}\] |
| III. | \[\ce{CH3 - \overset{⊕}{C}H - CH2 - OCH3}\] |
Match the intermediates given in Column I with their probable structure in Column II.
| Column I | Column II |
| (i) Free radical | (a) Trigonal planar |
| (ii) Carbocation | (b) Pyramidal |
| (iii) Carbanion | (c) Linear |
Which of the following carbocation's is most stable?
 |
 |
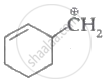 |
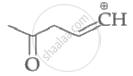 |
| (I) | (II) | (III) | (IV) |
Among the given species the resonance stabilised carbocations are:
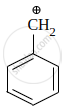 |
\[\ce{CH2 = \overset{⊕}{C}H}\] | \[\ce{CH3 - \overset{⊕}{C}H2}\] | \[\ce{HC ≡ \overset{⊕}{C}}\] |
| A | B | C | D |
The correct order of stability of given carbocation is:
The increasing order of stability of the following free radicals is ______.
