Advertisements
Advertisements
प्रश्न
Write the balanced chemical equation for an equilibrium reaction for which the equilibrium constant is given by expression.
`"K"_"C" = (["NH"_3]^4["O"_2]^5)/(["NO"]^4["H"_2"O"]^6)`
उत्तर
Balanced equation is:
\[\ce{4NO + 6H2O <=> 4NH3 + 7O2}\]
APPEARS IN
संबंधित प्रश्न
Which one of the following is incorrect statement ?
K1 and K2 are the equilibrium constants for the reactions respectively.
\[\ce{N2(g) + O2(g) <=>[K1] 2NO(g)}\]
\[\ce{NO(g) + O2(g) <=>[K2] 2NO2(g)}\]
What is the equilibrium constant for the reaction \[\ce{NO2(g) <=> 1/2 N2(g) + O2(g)}\]
In the equilibrium,
\[\ce{2A(g) <=> 2B(g) + C2(g)}\]
the equilibrium concentrations of A, B and C2 at 400 K are 1 × 10–4 M, 2.0 × 10–3 M, 1.5 × 10–4 M respectively. The value of KC for the equilibrium at 400 K is
\[\ce{[CO(H2O)6]^2+ (aq) (pink) + 4Cl- (aq) <=> [CoCl4]^2- (aq) (blue) + 6 H2O (l)}\]
In the above reaction at equilibrium, the reaction mixture is blue in colour at room temperature. On cooling this mixture, it becomes pink in color. On the basis of this information, which one of the following is true?
The equilibrium constants of the following reactions are:
\[\ce{N2 + 3H2 <=> 2NH3}\]; K1
\[\ce{N2 + O2 <=> 2NO}\]; K2
\[\ce{H2 + 1/2O2 <=> H2O}\]; K3
The equilibrium constant (K) for the reaction;
\[\ce{2NH3 + 5/2 O2 <=> 2NO + 3H2O}\], will be
For a given reaction at a particular temperature, the equilibrium constant has a constant value. Is the value of Q also constant? Explain.
For the reaction, \[\ce{A2(g) + B2(g) <=> 2AB(g); \Delta H}\] is -ve.
the following molecular scenes represent differenr reaction mixture. (A-green, B-blue)
| Closed ← |
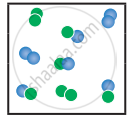 |
 |
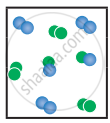 |
| System | At equilibrium | (x) | (y) |
- Calculate the equilibrium constant Kp and (Kc).
- For the reaction mixture represented by scene (x), (y) the reaction proceed in which directions?
- What is the effect of an increase in pressure for the mixture at equilibrium?
For the reaction
\[\ce{SrCO3(s) <=> SrO(s) + CO2(g)}\]
the value of equilibrium constant Kp = 2.2 × 10-4 at 1002 K. Calculate Kc for the reaction.
At particular temperature Kc = 4 × 10-2 for the reaction, \[\ce{H2S (g) <=> H2(g) +1/2 S2(g)}\]. Calculate the Kc for the following reaction.
\[\ce{3H2S (g) <=> 3H2 (g) + 3/2 S2 (g)}\]
The equilibrium constant Kp for the reaction \[\ce{N2 (g) + 3H2 (g) <=> 2NH3 (g)}\] is 8.19 × 102 at 298 K and 4.6 × 10-1 at 498 K. Calculate ∆H° for the reaction.
