Advertisements
Advertisements
प्रश्न
Write the names and formulas of the products formed when methane reacts with chlorine. Write the chemical equations.
उत्तर
Substitution reaction takes place.
Methane with Chlorine:
\[\ce{CH4 + Cl2 ->[Sunlight] \underset{Methyl chloride}{CH3Cl} + HCl}\]
or
1.
Methyl chloride
\[\begin{array}{cc}
\ce{H}\phantom{....................}\ce{H}\phantom{.......}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{H - C - H + Cl2 -> H - C - Cl + HCl}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{H}\phantom{....................}\ce{H}\phantom{.......}\\
\end{array}\]
2. Dichloro methane
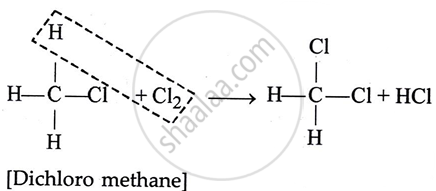
3. Chloroform or [Tri chlorornethane]
\[\begin{array}{cc}
\ce{Cl}\phantom{....................}\ce{Cl}\phantom{.......}\\
|\phantom{......................}|\phantom{.......}\\
\ce{Cl - C - H + Cl2 -> Cl - C - Cl + HCl}\\
|\phantom{......................}|\phantom{.......}\\
\ce{H}\phantom{.....................}\ce{H}\phantom{.......}\\
\end{array}\]
4. Carbon-tetrachloride
\[\begin{array}{cc}
\ce{Cl}\phantom{...................}\ce{Cl}\phantom{.......}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{Cl - C - H + Cl2 -> Cl - C - Cl + HCl}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{H}\phantom{....................}\ce{Cl}\phantom{.......}\\
\end{array}\]
संबंधित प्रश्न
Name the following:
The process by which ethane is obtained from ethane.
Match the options A to E with the statements
| A | Alkyne | 1 | No. of molecules in 22.4 dm-3 of carbon dioxide at STP |
| B | Alkane | 2 | An element with the electronic configuration 2,8,8,3 |
| C | Iron | 3 | CnH2n + 2 |
| D | 6.023 × 1023 | 4 | CnH2n − 2 |
| E | Metal | 5 | The metal which forms two types of ions |
Write the IUPAC and common names of these isomers
Write the:
electron dot formula
How is ethane prepared in the laboratory?
Write the names and the formula of the products formed when methane
Reacts with bromine Write the chemical equations
Write a balanced chemical equation for the preparation of :
Ethyne using calcium carbide
Write a balanced chemical equation for the preparation of :
Methane from sodium acetate
Identify the statement which does not describe the property of alkenes:
Which type of reaction will ethane undergo?
