Advertisements
Advertisements
प्रश्न
Write the names and formulas of the products formed when methane reacts with chlorine. Write the chemical equations.
उत्तर
Substitution reaction takes place.
Methane with Chlorine:
\[\ce{CH4 + Cl2 ->[Sunlight] \underset{Methyl chloride}{CH3Cl} + HCl}\]
or
1.
Methyl chloride
\[\begin{array}{cc}
\ce{H}\phantom{....................}\ce{H}\phantom{.......}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{H - C - H + Cl2 -> H - C - Cl + HCl}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{H}\phantom{....................}\ce{H}\phantom{.......}\\
\end{array}\]
2. Dichloro methane
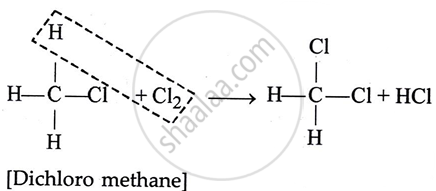
3. Chloroform or [Tri chlorornethane]
\[\begin{array}{cc}
\ce{Cl}\phantom{....................}\ce{Cl}\phantom{.......}\\
|\phantom{......................}|\phantom{.......}\\
\ce{Cl - C - H + Cl2 -> Cl - C - Cl + HCl}\\
|\phantom{......................}|\phantom{.......}\\
\ce{H}\phantom{.....................}\ce{H}\phantom{.......}\\
\end{array}\]
4. Carbon-tetrachloride
\[\begin{array}{cc}
\ce{Cl}\phantom{...................}\ce{Cl}\phantom{.......}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{Cl - C - H + Cl2 -> Cl - C - Cl + HCl}\\
|\phantom{.....................}|\phantom{.......}\\
\ce{H}\phantom{....................}\ce{Cl}\phantom{.......}\\
\end{array}\]
संबंधित प्रश्न
Classify the compound C3H8 as alkanes, alkenes, and alkynes.
Give the general formula of alkanes.
Draw the structures of the isomer of pentane. Write the IUPAC and common names of these isomer
How is methane prepared in the laboratory?
Name the compound prepared from ethyl bromide. Write a balanced equation for the same.
Identify the statement which does not describe the property of alkenes:
Draw the structure of isomers of butene and write their IUPAC names.
Write the equation for the following reaction:
Bromoethane and an aqueous solution of sodium hydroxide.
Give reasons:
Hydrocarbons are excellent fuels.
Write the IUPAC name of the following :
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C = C - C - H}\\
\phantom{.........}|\\
\phantom{.........}\ce{H}\\
\end{array}\]
