Advertisements
Advertisements
प्रश्न
Write the structures of the isomers of alcohols with molecular formula \[\ce{C4H10O}\]. Which of these exhibits optical activity?
उत्तर
The alcohol with molecular formula C4H10O is butanol and has 4 isomers, these are:
(a)
\[\ce{\underset{Butan-1-ol}{CH3 - CH2 - CH2 - CH2 - OH}}\]
(b)
\[\begin{array}{cc}
\phantom{}\ce{CH3 - \overset{∗}{C}H - CH2 - CH3}\\
\phantom{}|\phantom{.........}\\
\phantom{}\ce{\underset{Butan-2-ol}{OH}}\phantom{.......}
\end{array}\]
(c)
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - OH}\phantom{.}\\
\phantom{}|\phantom{.........}\\
\phantom{}\ce{\underset{2-Methylpropan-1-ol}{CH3}}\phantom{......}
\end{array}\]
(d)
\[\begin{array}{cc}
\phantom{...}\ce{CH3}\phantom{}\\
\phantom{}|\phantom{}\\
\phantom{}\ce{CH3 - C - CH3}\phantom{}\\
\phantom{}|\phantom{}\\
\phantom{...}\ce{\underset{2-methylpropan-2-ol}{OH}}\phantom{.}
\end{array}\]
Only butane-2-ol has chiral carbon due to it being the alcohol with molecular formula C4H10O exhibits optical activity.
APPEARS IN
संबंधित प्रश्न
Write the structure of the product of the following reaction:
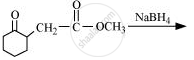
Write the structure of the product of the following reaction:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CHO ->[NaBH4]}\\
|\phantom{....}\\
\ce{CH3}\phantom{.}
\end{array}\]
Write the structure of the compound whose IUPAC name is as follows:
1-Phenylpropan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
3-Cyclohexylpentan-3-ol
Explain why is \[\ce{O = C =O}\] nonpolar while \[\ce{R - O - R}\] is polar.
Glycerol is used
Diethyl ether finds are in medicine as
Ether can be used
\[\begin{array}{cc}\phantom{......}\ce{OH}\\
\phantom{....}|\\\ce{CH3CH2 - C - CH3}\\
\phantom{....}|\\\phantom{.....}\ce{Ph}\end{array}\] cannot be prepared by ______.
Why is the C-O bond length in phenols less than that in methanol?
