Advertisements
Advertisements
प्रश्न
Zinc oxide reacts with carbon, on heating, to form zinc metal and carbon monoxide. Write a balanced chemical equation for this reaction. Name (i) oxidising agent, (ii) reducing agent, in this reaction.
उत्तर
The balanced chemical reaction is as follows:
ZnO + C → Zn + CO
(i) Oxidising agent: ZnO
It oxidises C to CO.
(ii) Reducing agent: C
It reduces ZnO to Zn.
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word:
Chemical equations are balanced to satisfy the law of ............
Write word equation for the following skeletal equation:
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write your observation for the following chemical reaction and name the product formed :
When dilute acetic acid is poured on baking soda.
Balance the following equation. Also name the product formed.
FeCI2 + CI2 → FeCI3
Balance the following equation:
HNO3 + H2S → NO2 + H2O + S
Balance the following simple equation:
Al + H2SO4 → Al2(SO4)3 + H2
Fill in the blank with the correct word from the options given below:
A chemical equation is a shorthand form for a _______ change.
Which among the following is(are) double displacement reaction(s)?
- `"Pb" + "CuCl"_2 -> "PbCl"_2 + "Cu"`
- `"Na"_2"SO"_4 + "BaCl"_2 -> "BaSO"_4 + 2"NaCl"`
- `"C" + "O"_2 -> "CO"_2`
- `"CH"_4 + 2"O"_2 -> "CO"_2 + 2"H"_2"O"`
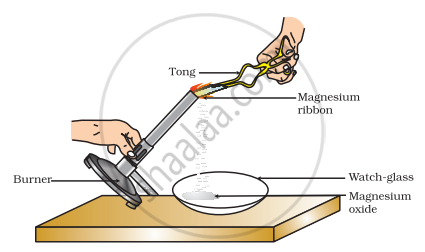
Which of the following is the correct observation of the reaction shown in the above set up?
Why should a magnesium ribbon be cleaned before burning in air?
