HSC Science (General)
HSC Science (Electronics)
HSC Science (Computer Science)
Academic Year: 2021-2022
Date: मार्च 2022
Advertisements
General instructions:
- The question paper is divided into four sections:
- Section A: Question No. 1 contains 10 multiple choice type questions carrying one mark each. Question No. 2 contains 8 very short answer type questions carrying one mark each.
- Section B: Question No. 3 to Question No. 14 are 12 short answer-I type questions carrying two marks each. Attempt any eight questions.
- Section C: Question No. 15 to Question No. 26 are 12 short answer-II type questions carrying two marks each. Attempt any eight questions.
- Section D: Question No. 27 to Question No. 31 are 5 long answer type questions carrying four marks each. Attempt any three questions.
- Start each section on a new page.
- Figures to the right indicate full marks.
- For each MCQ, correct answer must be written along with its alphabet.
e.g., (a) ..... / (b ) .... / ( c ) .... / ( d) ..... Only first attempt will be considered for evaluation. - Draw neat, labelled diagrams and write balanced chemical equations wherever necessary.
- Use of log table is necessary. Use of calculator is not allowed.
The following ion has the maximum number of unpaired electrons.
Sc3+
Ti3+
Fe3+
Co2+
Chapter: [0.08] Transition and Inner Transition Elements
The units of Henry’s law constant are ______.
bar dm3 mol–1
mol L–1 bar–1
L mol–1 bar–1
bar L–1 mol–1
Chapter: [0.02] Solutions
The following amines is the product of Gabriel phthalimide synthesis.
secondary aliphatic amine
primary aliphatic amine
aromatic primary amine
tertiary aliphatic amine
Chapter: [0.13] Amines [13.01] Amines
Sulfur dioxide reacts with sodium hydroxide solution to form _______.
sodium sulfite
sodium sulfate
sodium hydrogen sulfite
sodium hydrogen sulfate
Chapter: [0.07] Elements of Groups 16, 17 and 18 [7.04] Group 18 Elements
Ketones CANNOT be converted into carboxylic acid using _____________
Alkaline KMnO4
CrO3
dil. HNO3
hot and conc. HNO3
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids
The geometry of [CoF6]3– complex ion is _______.
Trigonal bipyramidal
Tetrahedral
Octahedral
square planar
Chapter: [0.09] Coordination Compounds
Which of the following is a biodegradable polymer?
nylon 6
nylon 6, 6
nylon 2-nylon 6
viscose rayon
Chapter: [0.15] Introduction to Polymer Chemistry
Aniline on reaction with bromine water produces ________________
1,4-Dibromobenzene
1,2,4-Tribromobenzene
2,4-Dibromoaniline
2,4,6-Tribromoaniline
Chapter: [0.13] Amines
A compound is made up of two elements X and Y and crystallizes in bcc structure. Atoms of X are present at the corners of the cube. Atoms of Y are present at the centre of the cube. The formula of the compound is ______.
X2Y
XY
XY2
X2Y3
Chapter: [0.01] Solid State [0.01] Solid State
For first order reaction, the rate constant for the decomposition of N2O5 is 6 × 10–4 s –1. The half-life period for decomposition in seconds is ______.
11.55
115.5
1155
1.155
Chapter: [0.06] Chemical Kinetics
Write the relationship between radius of atom and edge length of fcc unit cell.
Chapter: [0.01] Solid State
Label the one conjugate acid-base pair in the following reaction.
\[\ce{CO^{2-}_{3(aq)} + H2O_{(l)} ⇌ OH^- + HCO^-_3}\]
Chapter: [0.03] Ionic Equilibria
Draw intramolecular hydrogen bonding structure in o-nitrophenol.
Chapter: [0.11] Alcohols, Phenols and Ethers
What is Lanthanoid contraction?
Chapter: [8.02] F-block Elements
Write the order of ionic character of halides with monovalent metal (M).
Chapter: [0.07] Elements of Groups 16, 17 and 18
Advertisements
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids
Complete the following reaction.
\[\ce{H2N - CH2 - COOH + H2N - CH2 - CH2 - COOH ->[-H2O] ?}\]
Chapter: [0.14] Biomolecules
Calculate enthalpy of formation of HCl if bond enthalpies of H2, Cl2 and HCl are 434 kJ mol-1, 242 kJ mol–1 and 431 kJ mol–1 respectively.
Chapter: [0.04] Chemical Thermodynamics
Answer the following
How nanotechnology plays an important role in water purification techniques?
Chapter: [0.16] Green Chemistry and Nanochemistry
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Chapter: [0.13] Amines [13.01] Amines
Classify the following species into Lewis acids and Lewis bases.
| Cl- | |
| `"NH"_4^+` | |
| BCl3 | |
| NH3 |
Chapter: [0.03] Ionic Equilibria
In what way Kf and Kb are similar and in what way they are different?
Chapter: [0.02] Solutions
Derive an expression for pressure-volume work.
Chapter: [0.04] Chemical Thermodynamics
Describe the following:
Cannizzaro reaction
Chapter: [12.01] Aldehydes and Ketones
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
Chapter: [0.1] Halogen Derivatives
Write four points of distinction between Lanthanoids and Actinoids.
Chapter: [0.08] Transition and Inner Transition Elements [8.02] F-block Elements
Write chemical equation of acetyl chloride with phenol
Chapter: [0.11] Alcohols, Phenols and Ethers
Define atom economy.
Chapter: [0.16] Green Chemistry and Nanochemistry
The effective atomic number of cobalt in [Co(NH3)6]3+ is _____________
33
34
35
36
Chapter: [0.09] Coordination Compounds
For the reaction 2NOBr → 2NO2 + Br2, the rate law is rate = k[NOBr]2. If the rate of a reaction is 6.5 × 10–6 mol L–1 s–1, when the concentration of NOBr is 2 × 10–3 mol L–1. What would be the rate constant of the reaction?
Chapter: [0.06] Chemical Kinetics
Define the Standard enthalpy of combustion.
Chapter: [0.04] Chemical Thermodynamics
What is Grignard reagent? How is it prepared? Why are they prepared under anhydrous condition?
Chapter: [0.1] Halogen Derivatives
Advertisements
Answer the following.
Distinguish between rhombic sulfur and monoclinic sulfur.
Chapter: [0.07] Elements of Groups 16, 17 and 18
Compound ‘A’ with molecular formula C6H5Cl is fused with NaOH at high temperature under pressure to give compound ‘B’. Compound ‘B’ on treatment with dil.HCl gives compound C having characteristic carbolic odour. Write the chemical equations in support of this. Name the process and give uses of compound C.
Chapter: [0.11] Alcohols, Phenols and Ethers
Calculate the packing efficiency for bcc lattice.
Chapter: [0.01] Solid State
For a zero-order reaction, molecularity can never be equal to zero. Explain.
Chapter: [0.06] Chemical Kinetics
With the help of vapour pressure-temperature curves for solution and solvent, explain why boiling point of solvent is elevated when a nonvolatile solute is dissolved into it.
Chapter: [0.02] Solutions
Write three physical properties of sulfuric acid.
Chapter: [0.07] Elements of Groups 16, 17 and 18
Answer the following.
Explain in detail free radical mechanism involved during preparation of addition polymer.
Chapter: [0.15] Introduction to Polymer Chemistry
Draw a neat diagram for the following:
Haworth formula of maltose
Chapter: [0.14] Biomolecules
The solubility of AgBr in water is 1.20 × 10–5 mol dm–3. Calculate the solubility product of AgBr.
Chapter: [0.03] Ionic Equilibria
A buffer solution contains 0.3 mol dm–3 NH4OH (Kb = 1.8 × 10–5) and 0.4 mol dm–3 NH4Cl. Calculate pOH of the solution.
Chapter: [0.03] Ionic Equilibria
Write working of NICAD storage cell. Also write its applications.
Chapter: [0.05] Electrochemistry
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Ethane-1,2-diol in presence of dry HCl.
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids
Explain, why [Co(NH3)6]3+ ion is low spin? Calculate number of unpaired electrons and write the geometry of [Co(NH3)6]3+.
Chapter: [0.09] Coordination Compounds
Explain the trend in an Oxidation state of 3d elements.
Chapter: [0.08] Transition and Inner Transition Elements
Observe the following compounds and answer the questions given below.
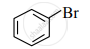
(I)
\[\ce{\underset{\text{(II)}}{CH3 - CH2 - Br}}\]
- Identify the type of halides.
- Explain the nature of the C – Br bond in both of these halides.
- Which of these compounds will undergo aqueous alkaline hydrolysis readily? Write the reaction in support of your answer.
Chapter: [0.1] Halogen Derivatives [10.01] Haloalkanes
Answer in brief.
How much heat is evolved when 12 g of CO reacts with NO2? The reaction is:
4CO(g) 2NO2(g) → 4CO2(g) + N2(g), ΔrH° = - 1200 kJ
Chapter: [0.04] Chemical Thermodynamics
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 12th Standard Board Exam Chemistry with solutions 2021 - 2022
Previous year Question paper for Maharashtra State Board 12th Standard Board Exam Chemistry-2022 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 12th Standard Board Exam.
How Maharashtra State Board 12th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
