Advertisements
Advertisements
Derive the relation between half life and rate constant for a first order reaction
Concept: Half Life Period of a Reaction
Write two factors that affect the rate of reaction.
Concept: Factors Influencing Rate of a Reaction
For a reaction : 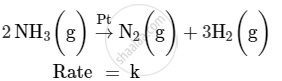
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Concept: Factors Influencing Rate of a Reaction
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
Concept: Temperature Dependence of the Rate of a Reaction
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
Concept: Factors Influencing Rate of a Reaction
A first order reaction takes 30 minutes for 50% completion. Calculate the time required for 90% completion of this reaction.
(log 2 = 0.3010)
Concept: Half Life Period of a Reaction
Define activation energy.
Concept: Temperature Dependence of the Rate of a Reaction
Define Reference electrode
Concept: Reference Electrodes
Derive the relation between half life and rate constant for a first order reaction
Concept: Half Life Period of a Reaction
In the extraction of Al, impure Al2O3 is dissolved in conc. NaOH to form sodium aluminate and leaving impurities behind. What is the name of this process?
Concept: Occurrence of Metals
A first order reaction takes 30 minutes for 50% completion. Calculate the time required for 90% completion of this reaction.
(log 2 = 0.3010)
Concept: Half Life Period of a Reaction
Explain the principle of the method of electrolytic refining of metals. Give one example.
Concept: Refining of Crude Metals
Derive an expression for the relation between half-life and rate constant for first-order reaction.
Concept: Integrated Rate Equations
Write four points of distinction between Lanthanoids and Actinoids.
Concept: F-block Elements > The Actinoids
The following electronic configuration of elements shows highest oxidation state
Concept: Properties of Lanthanoids
Write any two uses of actinides.
Concept: Applications of Actinoids
Write the IUPAC name of the given compound:
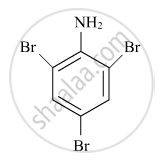
Concept: Nomenclature of Coordination Compounds - Naming of Mononuclear Coordination Compounds
When a co-ordination compound CrCl3.6H2O is mixed with AgNO3, 2 moles of AgCl are precipitated per mole of the compound. Write
(i) Structural formula of the complex.
(ii) IUPAC name of the complex.
Concept: Nomenclature of Coordination Compounds - Naming of Mononuclear Coordination Compounds
Write down the IUPAC name of the following complex: [Cr(NH3)2Cl2(en)]Cl (en = ethylenediamine)
Concept: Nomenclature of Coordination Compounds - Naming of Mononuclear Coordination Compounds
Draw the geometrical isomers of complex [Pt(NH3)2Cl2].
Concept: Isomerism in Coordination Compounds > Stereoisomerism
