Advertisements
Advertisements
प्रश्न
A beam of monochromatic radiation is incident on a photosensitive surface. Answer the following question giving reason :
Do the emitted photoelectrons have the same kinetic energy?
उत्तर
Yes, all the emitted photoelectrons have the same kinetic energy as the kinetic energy of the emitted photoelectrons depends upon the frequency of the incident radiation for a given photosensitive surface.
APPEARS IN
संबंधित प्रश्न
Write three characteristic features in photoelectric effect that cannot be explained on the basis of wave theory of light, but can be explained only using Einstein's equation.
Light of intensity ‘I’ and frequency ‘v’ is incident on a photosensitive surface and causes photoelectric emission. What will be the effect on anode current when (i) the intensity of light is gradually increased. In each case, all other factors remain the same. Explain, giving justification in each case.
Two monochromatic beams, one red and the other blue, have the same intensity. In which case (i) the number of photons per unit area per second is larger, (ii) the maximum kinetic energy of the photoelectrons is more? Justify your answer.
Draw a plot showing the variation of photoelectric current versus the intensity of incident radiation on a given photosensitive surface.
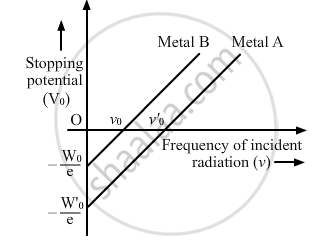
The graph shows the variation of stopping potential with frequency of incident radiation for two photosensitive metals A and B. Which one of the two has higher value of work-function? Justify your answer.
The work functions for potassium and caesium are 2.25 eV and 2.14 eV respectively. Is the photoelectric effect possible for either of them if the incident wavelength is 5180 Å?
[Given : Planck’s constant = 6.63 x 10–34 J.s.;
Velocity of light = 3 x 108 m/s; 1 eV = 1.6 x 10–19 J]
In an experiment of the photoelectric effect, the graph of maximum kinetic energy EK of the emitted photoelectrons versus the frequency v of the incident light is a straight line AB shown in Figure 6 below:
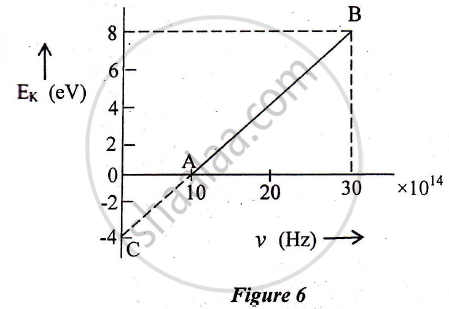
Find:
1) Threshold frequency of the metal
2) The work function of the metal.
3) Stopping potential for the photoelectrons emitted by the light of frequency `v = 30 xx 10^14 Hz`
Draw a plot showing the variation of photoelectric current with collector potential for different frequencies but same intensity of incident radiation ?
Plot a labelled graph of IVsl where Vs is stopping potential versus frequency f of the incident radiation.
Light of wavelength 4000 Å is incident on two metals A and B. Which metal will emit photoelectrons, if their work functions are 3.8 e V and 1.6 e V respectively?
