Advertisements
Advertisements
प्रश्न
A compound 'X' on heating with excess conc. sulphuric acid at 443 K gives an unsaturated compound 'Y'. 'X' also reacts with sodium metal to evolve a colourless gas 'Z'. Identify 'X', 'Y' and 'Z'. Write the equation of the chemical reaction of formation of 'Y' and also write the role of sulphuric acid in the reaction.
उत्तर
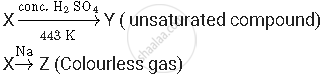
X, Y and Z are alcohol, alkene and hydrogen respectively.
Suppose X is ethanol, it gives alkene on reacting with conc. H2SO4

Role of sulphuric acid: It is used as a dehydrating agent in the above reaction.
APPEARS IN
संबंधित प्रश्न
Define Saturated hydrocarbon.
Explain in brief two main reasons for carbon forming a large number of compounds.
Draw the electron dot structures for ethanoic acid.
Give a test that can be used to differentiate chemically between butter and cooking oil.
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethyne
An unsaturated hydrocarbon having a triple covalent bond has 50 hydrogen atoms in its molecule. The number of carbon atoms in its molecule will be:
(a) 24
(b) 25
(c) 26
(d) 28
Fill in the blanks and rewrite the completed statements:
The organic compounds having double or triple bond in them are termed as _________________ _________________.
Structural formula of ethyne is ______.
Structural formula of ethyne is
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\ce{CH3}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
