Advertisements
Advertisements
प्रश्न
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethyne
उत्तर
Structure of ethyne (C2H2) is as follows:
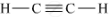
Ethyne has one triple bond between its carbon atoms, and two single bonds between its carbon and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
A compound 'X' on heating with excess conc. sulphuric acid at 443 K gives an unsaturated compound 'Y'. 'X' also reacts with sodium metal to evolve a colourless gas 'Z'. Identify 'X', 'Y' and 'Z'. Write the equation of the chemical reaction of formation of 'Y' and also write the role of sulphuric acid in the reaction.
Write the electron-dot structures for ethene.
Write the electron-dot structures for ethyne.
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethene
Give the names and structural formulae of one saturated cyclic hydrocarbon and one unsaturated cyclic hydrocarbon.
Out of the following pairs of compounds, the unsaturated compounds are:
(a) C2H6 and C4H6
(b) C6H12 and C5H12
(c) C4H6 and C6H12
(d) C2H6 and C4H10
Structural formula of ethyne is ______.
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\ce{CH3}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
Name the functional groups present in the following compounds
- CH3 CO CH2 CH2 CH2 CH3
- CH3 CH2 CH2 COOH
- CH3 CH2 CH2 CH2 CHO
- CH3 CH2 OH
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
