Advertisements
Advertisements
प्रश्न
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
benzene
उत्तर
Structure of benzene (C6H6 ) is as follows:
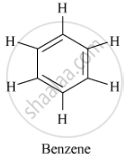
A benzene molecule has three carbon-carbon double bonds, three carbon-carbon single bonds and six carbon-hydrogen single bonds.
APPEARS IN
संबंधित प्रश्न
What is meant by isomers?
Give reason why the carbon compounds generally have low melting and boiling points.
What are hydrocarbons? Explain with examples.
Explain the meaning of saturated and unsaturated hydrocarbons with two examples each.
Two organic compounds A and B have the same molecular formula C6H12. Write the names and structural formulae:
if A is a cyclic compound
Two organic compounds A and B have the same molecular formula C6H12. Write the names and structural formulae:
Which compound contains only single bonds?
Write the molecular and structural formula of a cyclic hydrocarbon whose molecule contains 8 atoms of carbon.
A reagent which can help us to distinguish between alkenes and alkynes is ______.
How many isomers are possible for the compound with the molecular formula C4H8? Draw the electron dot structure of branched-chain isomer.
Draw the structure of propanone.
