Advertisements
Advertisements
प्रश्न
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
benzene
उत्तर
Structure of benzene (C6H6 ) is as follows:
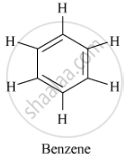
A benzene molecule has three carbon-carbon double bonds, three carbon-carbon single bonds and six carbon-hydrogen single bonds.
APPEARS IN
संबंधित प्रश्न
Define the term ‘structural’ isomerism'.
List two reasons for carbon forming a large number of compounds. Name the type of bonding found in most of its compounds. Why does carbon form compounds mainly by this kind of bonding?
Draw the structures for Butanone
Explain why propane cannot exhibit the structural isomerism property.
Write the molecular formula and structure of benzene.
The pair of elements which exhibits the property of catenation is:
The solid element A exhibits the property of catenation. It is also present in the form of a gas B in the air which is utilised by plants in photosynthesis. An allotrope C of this element is used in glass cutters.
(a) What is element A?
(b) What is the gas B?
(c) Name the allotrope C.
(d) State another use of allotrope C (other than in glass cutters).
(e) Name another allotrope of element A which exists as spherical molecules.
(f) Name a yet another allotrope of element A which conduct electricity.
You are given the following molecular formulae of some hydrocarbons:
Which formula represents benzene?
How many isomers are possible for the compound with the molecular formula C4H8? Draw the electron dot structure of branched-chain isomer.
Write the structures of two compounds having molecular formula C3H6O and give their names.
