Advertisements
Advertisements
प्रश्न
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethene
उत्तर
Structure of ethene (C2H4) is as follows:
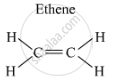
Ethene has a double bond between its carbon atoms, and four single bonds between its carbon and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
Give any two differences between alkanes and alkenes.
Define Saturated hydrocarbon.
Draw the electron dot structures for ethanoic acid.
Give a test that can be used to differentiate chemically between butter and cooking oil.
Write the electron-dot structures for ethane.
Write the molecular formula of an alkene having 20 carbon atoms?
A mixture of ethyne (acetylene) and oxygen is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
A compound X is formed by the reaction of a carboxylic acid C2H4O2 and an alcohol in presence of a few drops of H2SO4. The alcohol on oxidation with alkaline KMnO4 followed by acidification gives the same carboxylic acid as used in this reaction. Give the names and structures of
- carboxylic acid
- alcohol and
- the compound X.
Also write the reaction.
What is the role of metal or reagents written on arrows in the given chemical reactions?
- \[\begin{array}{cc}
\ce{CH3}\phantom{.....}\ce{CH3}\phantom{.........}\ce{CH3}\phantom{..}\ce{CH3}\phantom{..........}\\
\phantom{...}\backslash\phantom{......}/\phantom{................}|\phantom{.....}|\phantom{............}\\
\ce{C} = \ce{C} + \ce{H2} \overset{\ce{Ni}}{\rightarrow} \ce{CH3 - C - C - CH3}\\
\phantom{.}/\phantom{........}\backslash\phantom{..............}|\phantom{....}|\phantom{.........}\\
\ce{CH3}\phantom{....}\ce{CH3}\phantom{............}\ce{H}\phantom{...}\ce{H}\phantom{........}
\end{array}\] - \[\ce{CH3COOH + CH3CH2OH \overset{\ce{Conc. H2SO4}}{\rightarrow}CH3COOC2H5 + H2O}\]
- \[\ce{CH3CH2OH}\ce{->[Alk.KMnO4][Heat]CH3COOH}\]
Draw the electron dot structure for ethyne.
