Advertisements
Advertisements
प्रश्न
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethene
उत्तर
Structure of ethene (C2H4) is as follows:
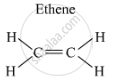
Ethene has a double bond between its carbon atoms, and four single bonds between its carbon and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
Define Saturated hydrocarbon.
Give a chemical test to distinguish between saturated and unsaturated hydrocarbons.
Draw the electron dot structures for F2.
Write the electron-dot structures for ethane.
Name two catalysts which can be used in the hydrogenation of unsaturated compounds.
A mixture of ethyne (acetylene) and oxygen is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Explain the following reaction with balanced chemical equation:
Calcium reacts with water.
Name the functional groups present in the following compounds
- CH3 CO CH2 CH2 CH2 CH3
- CH3 CH2 CH2 COOH
- CH3 CH2 CH2 CH2 CHO
- CH3 CH2 OH
- What are hydrocarbons? Give examples.
- Give the structural differences between saturated and unsaturated hydrocarbons with two examples each.
- What is a functional group? Give examples of four different functional groups.
- Write the name and draw the structure of a saturated hydrocarbon with four carbon atoms.
- Write the number of single covalent bonds present in this compound.
