Advertisements
Advertisements
Question
A compound 'X' on heating with excess conc. sulphuric acid at 443 K gives an unsaturated compound 'Y'. 'X' also reacts with sodium metal to evolve a colourless gas 'Z'. Identify 'X', 'Y' and 'Z'. Write the equation of the chemical reaction of formation of 'Y' and also write the role of sulphuric acid in the reaction.
Solution
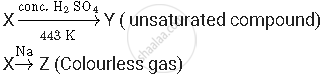
X, Y and Z are alcohol, alkene and hydrogen respectively.
Suppose X is ethanol, it gives alkene on reacting with conc. H2SO4

Role of sulphuric acid: It is used as a dehydrating agent in the above reaction.
APPEARS IN
RELATED QUESTIONS
Define Catenation.
What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Draw the electron dot structures for H2S.
Draw the electron dot structures for propanone.
Give a test that can be used to differentiate chemically between butter and cooking oil.
Give the names and structural formulae of one saturated cyclic hydrocarbon and one unsaturated cyclic hydrocarbon.
Write the molecular formula of an alkene having 20 carbon atoms?
Name two catalysts which can be used in the hydrogenation of unsaturated compounds.
Write the name of the following compound
\[\begin{array}{cc}
\phantom{....}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{O}\phantom{......}\\
\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}||\phantom{......}\\
\ce{H} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{OH}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.......}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\\
\end{array}\]
Write the name of the following compound
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - C - C - C = O}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.....}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}
\end{array}\]
