Advertisements
Advertisements
प्रश्न
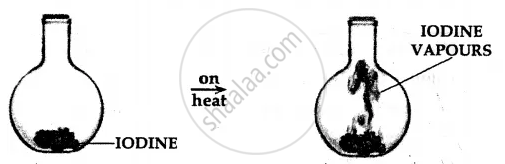
A crystal of iodine is placed in a closed flat bottom flask and heated. State how you would conclude from the observations seen, that interparticle space is minimum in solids and maximum in gases.
उत्तर
Procedure:
A crystal of iodine is heated in a closed flask. The crystal containing ‘X’ no. of particles turns into vapours, which also contain ‘X’ no. of particles and fills the complete flask.
Conclusion:
- Particles of a solid [e.g. crystal of iodine] are more closely packed.
- Particles of a gas [e.g. vapours of iodine] are least closely packed and fill the flask completely.
APPEARS IN
संबंधित प्रश्न
Fill in the blank:
The change of water vapour into water is called ......................
When sodium chloride is added to a definite volume of water and stirred well, a solution is formed, but there is no increase in the level of water. Why?
Write your observation and conclusion for the following.
When a small stone is gently dipped into a glass filled with water.
Give reason Gases can be compressed easily .
Give reason of Why do liquids and gases flow but solids do not ?
Fill in the blank
When a gas is cooled, its molecules ________ energy,
There are large intermolecular gaps in
A measuring cylinder is filled with water to a particular mark. A piece of solid is immersed inside the measuring cylinder. State why the level of water in the measuring cylinder will rise up. If the solid is removed, what will be the new level of the water in the measuring cylinder? Give a reason for your answer.
Which one of the following element name is not derived from planet?
The property to flow is unique to fluids. Which one of the following statements is correct?
