Advertisements
Advertisements
Question
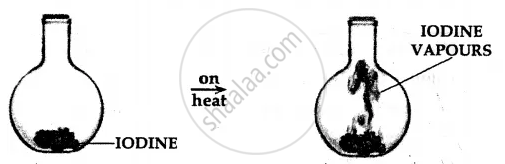
A crystal of iodine is placed in a closed flat bottom flask and heated. State how you would conclude from the observations seen, that interparticle space is minimum in solids and maximum in gases.
Solution
Procedure:
A crystal of iodine is heated in a closed flask. The crystal containing ‘X’ no. of particles turns into vapours, which also contain ‘X’ no. of particles and fills the complete flask.
Conclusion:
- Particles of a solid [e.g. crystal of iodine] are more closely packed.
- Particles of a gas [e.g. vapours of iodine] are least closely packed and fill the flask completely.
APPEARS IN
RELATED QUESTIONS
What do you observe when a gas jar which appears empty is inverted over a gas jar containing Bromine vapours? Name the phenomenon.
Classify the following into solids, liquids and gases.
Oxygen, milk, common salt, wax, stone, water vapour, carbon-dioxide, sugar, mercury, coal, blood, butter, copper, coconut oil, kerosene.
Why do solids, liquids and gases differ in their physical states ?
Define interconversion of states of matter.
The state of matter which has no definite shape or volume is called
- solid
- liquid
- gas
- water
Give reason
When an empty gas jar is inverted over a gas jar containing a coloured gas, the gas also spreads into the empty jar.
Fill in the following blank with suitable words :
The smell of perfume gradually spreads across a room due to .....................
Give a reason why:
Gases have no definite volume and no definite shape.
Fill in the blank:
Particles in a gas possess ___________ kinetic energy.
State in which of the following examples ie. a piece of wood, water, light gas is the Inter-particle space maximum.
