Advertisements
Advertisements
Question
Describe simple experiment to show that particles of matter are closely packed in solids and less in liquids.
Solution
Experiment – To show that particles are closely packed in solids and less in liquids
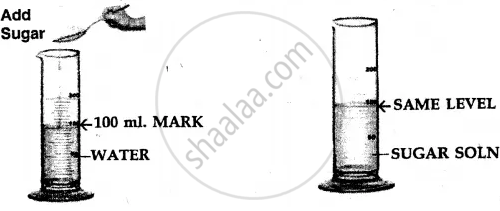
Procedure –
- Take a measuring cylinder and add 100 ml. of water to it.
- Add crystals of sugar [solid] to the water and stir carefully to obtain a sugar solution.
- The volume of water after the addition of sugar remains the
same i.e. at 100 ml. mark.
Conclusion –
- Particles of a liquid [e.g. water] are less closely packed.
- Particles of a solid [e.g. sugar] are closely packed.
- The sugar particles fill the intermolecular spaces, which must be existing between the water molecules, and hence the level of water in the measuring cylinder does not rise.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Give reasons:
A gas can fill the whole vessel in which it is enclosed.
Define interconversion of states of matter.
Give reason for the following.
Pure substances have fixed melting or boiling points.
Fill in the blank:
Water is a matter because it has mas and occupies....................
Write whether the following statement is true or false.
Solids have the largest inter-molecular space.
Give reason of Why do liquids and gases flow but solids do not ?
Explain the following :
When an incense stick is lighted in the corner of a room, its fragrance spreads quickly in the entire room.
A glass beaker is half-filled with water and an empty glass tumbler is inverted & lowered inside the glass beaker. State your observations on tilting the tumbler below the level of the water in the glass beaker. Give a reason for your answer.
With reference to the liquid state of matter – answer the following
How does the stored energy, have an effect on inter-particles space.
