Advertisements
Advertisements
प्रश्न
Describe simple experiment to show that particles of matter are closely packed in solids and less in liquids.
उत्तर
Experiment – To show that particles are closely packed in solids and less in liquids
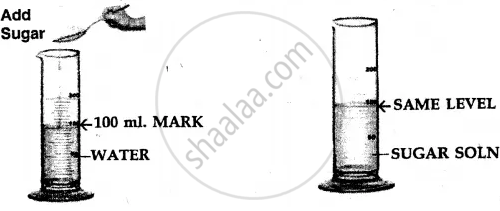
Procedure –
- Take a measuring cylinder and add 100 ml. of water to it.
- Add crystals of sugar [solid] to the water and stir carefully to obtain a sugar solution.
- The volume of water after the addition of sugar remains the
same i.e. at 100 ml. mark.
Conclusion –
- Particles of a liquid [e.g. water] are less closely packed.
- Particles of a solid [e.g. sugar] are closely packed.
- The sugar particles fill the intermolecular spaces, which must be existing between the water molecules, and hence the level of water in the measuring cylinder does not rise.
APPEARS IN
संबंधित प्रश्न
Name the three states of matter and define them.
Give reason:
Why do liquids and gases flow but solids do not ?
Name the phenomenon which cause the following change:
Disappearance of camphor.
When a stone is dipped in a glass containing some water, the level of water rises but when a spoon of sugar is added to it and stirred, the water level does not rise?
In which of the following conditions, the distance between the molecules of hydrogen gas would increase?
- increasing pressure on hydrogen contained in a closed container
- some hydrogen gas leaking out of the container
- increasing the volume of the container of hydrogen gas
- adding more hydrogen gas to the container without increasing the volume of the container
Define: interconversion of states of matter.
Name the three states of matter.
Differentiate the general properties of solids, liquids and gases in the form of a table — with reference to free diffusion.
State in which of the following examples ie. a piece of wood, water, light gas is the Energy possessed by particles of matter, very large.
Mention any two characteristics of the particles of matter.
