Advertisements
Advertisements
प्रश्न
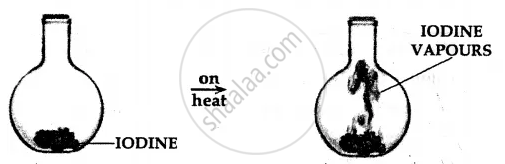
A crystal of iodine is placed in a closed flat bottom flask and heated. State how you would conclude from the observations seen, that interparticle space is minimum in solids and maximum in gases.
उत्तर
Procedure:
A crystal of iodine is heated in a closed flask. The crystal containing ‘X’ no. of particles turns into vapours, which also contain ‘X’ no. of particles and fills the complete flask.
Conclusion:
- Particles of a solid [e.g. crystal of iodine] are more closely packed.
- Particles of a gas [e.g. vapours of iodine] are least closely packed and fill the flask completely.
APPEARS IN
संबंधित प्रश्न
Give reasons:
A gas can fill the whole vessel in which it is enclosed.
Give two example of the following:
The substances which sublime.
Give two example of the following:
The substances which do not change their state on heating.
When sodium chloride is added to a definite volume of water and stirred well, a solution is formed, but there is no increase in the level of water. Why?
How will you prove by an experiment that solids occupy space ?
Write whether the following statement is true or false.
Only water can exist in three different states.
For the following statement, say whether it describe a solid, a liquid or a gas.
Particles are quite close together.
Name the phenomenon which cause the following change:
Formation of water vapour from water.
Fill in the blank
When a gas is cooled, its molecules ________ energy,
With reference to the liquid state of matter – answer the following
How does the stored energy, have an effect on inter-particles space.
