Advertisements
Advertisements
प्रश्न
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
उत्तर
Given: T1/2 = 30 min, T1 = 300 K
T1/2 = 10 min, T2 = 320 K
Formula: `log "K"_2/"K"_1 = "E"_"a"/(2.303 "R") [1/"T"_1 - 1/"T"_2]`
K = `0.693/("t"_(1//2))`
K1 at 300 K = `0.693/30` min−1
K2 at 320 K = `0.693/10` min−1
`log ("K"_2/"K"_1) = "E"_"a"/(2.303 "R") [1/"T"_1 - 1/"T"_2]`
`log ((0.693/10)/(0.693/30)) = "E"_"a"/(2.303 "R") [1/300 - 1/320]`
log (3) = `"E"_"a"/(2.303 "R") [20/(300 xx 320)]`
Ea = 0.4771 × 2.303 × 8.314 × 300 × 16
Ea = 43.8 kJ
APPEARS IN
संबंधित प्रश्न
Explain a graphical method to determine activation energy of a reaction.
Consider the reaction
`3I_((aq))^-) +S_2O_8^(2-)->I_(3(aq))^-) + 2S_2O_4^(2-)`
At particular time t, `(d[SO_4^(2-)])/dt=2.2xx10^(-2)"M/s"`
What are the values of the following at the same time?
a. `-(d[I^-])/dt`
b. `-(d[S_2O_8^(2-)])/dt`
c. `-(d[I_3^-])/dt`
The rate constant of a first order reaction increases from 4 × 10−2 to 8 × 10−2 when the temperature changes from 27°C to 37°C. Calculate the energy of activation (Ea). (log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
The decomposition of hydrocarbon follows the equation k = `(4.5 xx 10^11 "s"^-1) "e"^(-28000 "K"//"T")`
Calculate Ea.
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 x 1013 sec-1 and 98.6 kJ mol-1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K-1 mol-1]
Calculate activation energy for a reaction of which rate constant becomes four times when temperature changes from 30 °C to 50 °C. (Given R = 8.314 JK−1 mol−1).
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
Predict the main product of the following reactions: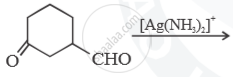
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
