Advertisements
Advertisements
प्रश्न
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
उत्तर
\[\ce{ C_12 H22O11 + H2O ->[H+] C6 + \underset{Glucose}{C6H12O6} + \underset{Fructose}{C6H12O6} }\] Rate=K[C12H22O4]
Biomolecular reaction is Kinetically first order. Consider a chemical reaction between two substances when one reactanct is present in large excess.
`CH_3 COOC_2H5 +H_2O -> CH_3OOH + C_2H_5OH`
t=0 0.01mol 10mol 0 0
t 0mol 9.9mol 0.01 0.01 mol
Rate = K[CH3COOC2H5]
`Rate = K[CH_3COOC_2H_5] where K=K' [H2O]` the reaction behaves as first order reactions.
APPEARS IN
संबंधित प्रश्न
The rate constant of a first order reaction increases from 2 × 10−2 to 4 × 10−2 when the temperature changes from 300 K to 310 K. Calculate the energy of activation (Ea).
(log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 x 1013 sec-1 and 98.6 kJ mol-1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K-1 mol-1]
Calculate activation energy for a reaction of which rate constant becomes four times when temperature changes from 30 °C to 50 °C. (Given R = 8.314 JK−1 mol−1).
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
Activation energy of a chemical reaction can be determined by ______.
Mark the incorrect statements:
(i) Catalyst provides an alternative pathway to reaction mechanism.
(ii) Catalyst raises the activation energy.
(iii) Catalyst lowers the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
Why in the redox titration of \[\ce{KMnO4}\] vs oxalic acid, we heat oxalic acid solution before starting the titration?
The slope of Arrhenius Plot `("In" "k" "v"//"s" 1/"T")` of first-order reaction is −5 × 103 K. The value of Ea of the reaction is. Choose the correct option for your answer. [Given R = 8.314 JK−1mol−1]
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
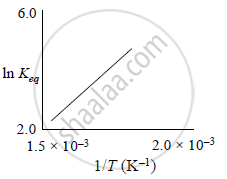
The reaction must be:
