Advertisements
Advertisements
प्रश्न
Mark the incorrect statements:
(i) Catalyst provides an alternative pathway to reaction mechanism.
(ii) Catalyst raises the activation energy.
(iii) Catalyst lowers the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
उत्तर
(ii) Catalyst raises the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
Explanation:
(i) As the catalyst is added to the reaction medium rate of reaction increases by decreasing activation energy of molecule. Hence, it follows an alternative pathway.
(ii) Catalyst does not change the enthapy change of reaction. Energy of ‘reactant and product remain same in both catalysed and uncatalysed reaction.
APPEARS IN
संबंधित प्रश्न
Consider the reaction
`3I_((aq))^-) +S_2O_8^(2-)->I_(3(aq))^-) + 2S_2O_4^(2-)`
At particular time t, `(d[SO_4^(2-)])/dt=2.2xx10^(-2)"M/s"`
What are the values of the following at the same time?
a. `-(d[I^-])/dt`
b. `-(d[S_2O_8^(2-)])/dt`
c. `-(d[I_3^-])/dt`
What will be the effect of temperature on rate constant?
The rate constant for the decomposition of hydrocarbons is 2.418 × 10−5 s−1 at 546 K. If the energy of activation is 179.9 kJ/mol, what will be the value of pre-exponential factor?
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
Consider figure and mark the correct option.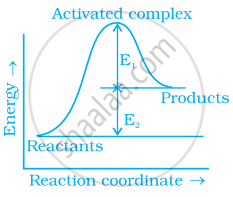
Oxygen is available in plenty in air yet fuels do not burn by themselves at room temperature. Explain.
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
In respect of the eqn k = \[\ce{Ae^{{-E_a}/{RT}}}\] in chemical kinetics, which one of the following statement is correct?
Arrhenius equation can be represented graphically as follows:

The (i) intercept and (ii) slope of the graph are:
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
