Advertisements
Advertisements
प्रश्न
Consider figure and mark the correct option.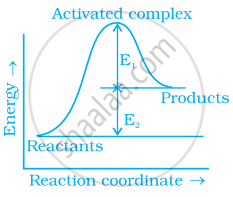
पर्याय
Activation energy of forward reaction is E1 + E2 and product is less stable than reactant.
Activation energy of forward reaction is E1 + E2 and product is more stable than reactant.
Activation energy of both forward and backward reaction is E1 + E2 and reactant is more stable than product.
Activation energy of backward reaction is E1 and product is more stable than reactant.
उत्तर
Activation energy of forward reaction is E1 + E2 and product is less stable than reactant.
Explanation:
Ea (forward) = E1 + E2
Since energy of reactants is less than products and the product is less stable than the reactant.
APPEARS IN
संबंधित प्रश्न
The rate constant of a first order reaction increases from 4 × 10−2 to 8 × 10−2 when the temperature changes from 27°C to 37°C. Calculate the energy of activation (Ea). (log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
What will be the effect of temperature on rate constant?
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/"T"` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
Total number of vibrational degrees of freedom present in CO2 molecule is
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
