Advertisements
Advertisements
प्रश्न
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
उत्तर
Thermodynamics feasibility of a reaction depends on Gibbs free energy i.e., AG must be negative for spontaneous process. Kinetic feasibility depends on the activation energy of reaction, the lesser is the activation energy, the greater is the feasibility of reaction, i.e.,
i.e., Diamond `->` Graphic ΔG = – ve
This process is thermodynamically feasible but it is very slow due to its high activation energy.
APPEARS IN
संबंधित प्रश्न
Consider the reaction
`3I_((aq))^-) +S_2O_8^(2-)->I_(3(aq))^-) + 2S_2O_4^(2-)`
At particular time t, `(d[SO_4^(2-)])/dt=2.2xx10^(-2)"M/s"`
What are the values of the following at the same time?
a. `-(d[I^-])/dt`
b. `-(d[S_2O_8^(2-)])/dt`
c. `-(d[I_3^-])/dt`
The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate Ea.
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
Calculate activation energy for a reaction of which rate constant becomes four times when temperature changes from 30 °C to 50 °C. (Given R = 8.314 JK−1 mol−1).
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
Activation energy of a chemical reaction can be determined by ______.
Total number of vibrational degrees of freedom present in CO2 molecule is
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
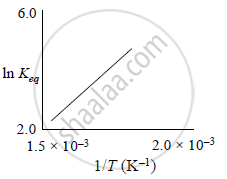
The reaction must be:
It is generally observed that the rate of a chemical reaction becomes double with every 10oC rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298K to 308K, what would be the value of activation energy (Ea) for the reaction?
