Advertisements
Advertisements
प्रश्न
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
उत्तर
Thermodynamics feasibility of a reaction depends on Gibbs free energy i.e., AG must be negative for spontaneous process. Kinetic feasibility depends on the activation energy of reaction, the lesser is the activation energy, the greater is the feasibility of reaction, i.e.,
i.e., Diamond `->` Graphic ΔG = – ve
This process is thermodynamically feasible but it is very slow due to its high activation energy.
APPEARS IN
संबंधित प्रश्न
The decomposition of hydrocarbon follows the equation k = `(4.5 xx 10^11 "s"^-1) "e"^(-28000 "K"//"T")`
Calculate Ea.
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
Predict the main product of the following reactions: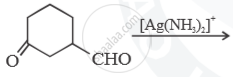
Consider figure and mark the correct option.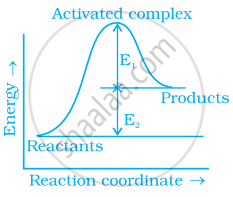
Why does the rate of a reaction increase with rise in temperature?
Why in the redox titration of \[\ce{KMnO4}\] vs oxalic acid, we heat oxalic acid solution before starting the titration?
The rate constant for a reaction is 1.5 × 10–7 sec–1 at 50°C. What is the value of activation energy?
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
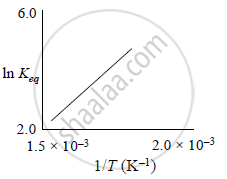
The reaction must be:
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
