Advertisements
Advertisements
प्रश्न
Predict the main product of the following reactions: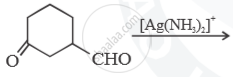
उत्तर
Predict main product of following reaction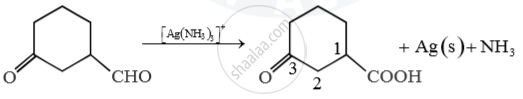
Main product 3-oxocyclohexanecarboxylic acid
APPEARS IN
संबंधित प्रश्न
Explain a graphical method to determine activation energy of a reaction.
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
What will be the effect of temperature on rate constant?
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
Consider figure and mark the correct option.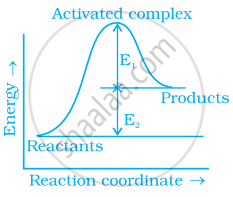
The reaction between \[\ce{H2(g)}\] and \[\ce{O2(g)}\] is highly feasible yet allowing the gases to stand at room temperature in the same vessel does not lead to the formation of water. Explain.
The activation energy in a chemical reaction is defined as ______.
The equation k = `(6.5 xx 10^12 "s"^(-1))"e"^(- 26000 " K"//"T")` is followed for the decomposition of compound A. The activation energy for the reaction is ______ kJ mol-1. (Nearest integer) (Given: R = 8.314 JK-1 mol-1)
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
It is generally observed that the rate of a chemical reaction becomes double with every 10oC rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298K to 308K, what would be the value of activation energy (Ea) for the reaction?
