Advertisements
Advertisements
प्रश्न
Why in the redox titration of \[\ce{KMnO4}\] vs oxalic acid, we heat oxalic acid solution before starting the titration?
उत्तर
The reaction between \[\ce{KMnO4}\] and oxalic acid is very slow. By raising the temperature we can increase the rate of reaction.
APPEARS IN
संबंधित प्रश्न
The rate constant of a first order reaction increases from 4 × 10−2 to 8 × 10−2 when the temperature changes from 27°C to 37°C. Calculate the energy of activation (Ea). (log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
The rate constant for the decomposition of hydrocarbons is 2.418 × 10−5 s−1 at 546 K. If the energy of activation is 179.9 kJ/mol, what will be the value of pre-exponential factor?
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 x 1013 sec-1 and 98.6 kJ mol-1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K-1 mol-1]
Explain the following terms :
Half life period of a reaction (t1/2)
Which of the following graphs represents exothermic reaction?
(a)
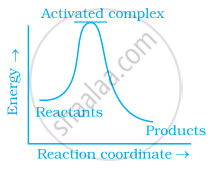
(b)
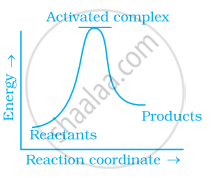
(c)
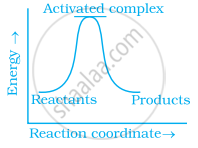
During decomposition of an activated complex:
(i) energy is always released
(ii) energy is always absorbed
(iii) energy does not change
(iv) reactants may be formed
The reaction between \[\ce{H2(g)}\] and \[\ce{O2(g)}\] is highly feasible yet allowing the gases to stand at room temperature in the same vessel does not lead to the formation of water. Explain.
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be
The activation energy in a chemical reaction is defined as ______.
