Advertisements
Advertisements
प्रश्न
Which of the following graphs represents exothermic reaction?
(a)
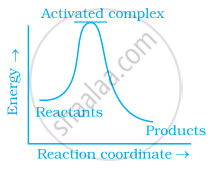
(b)
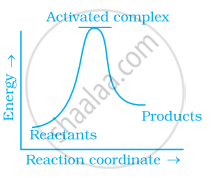
(c)
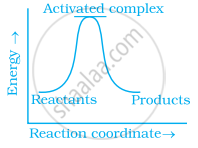
विकल्प
(a) only
(b) only
(c) only
(a) and (b)
उत्तर
(a) only
Explanation:
Exothermic reaction is a chemical reaction in which the enthalpy change is negative and energy is released in the form of light and heat.
As we all know, ΔH is negative.
Activation energy (forward reaction) – (backward reaction)
(Forward reaction) – Ea (backward reaction)
The product's exothermic energy exceeds the activation energy of the reactant.
APPEARS IN
संबंधित प्रश्न
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 x 1013 sec-1 and 98.6 kJ mol-1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K-1 mol-1]
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
Mark the incorrect statements:
(i) Catalyst provides an alternative pathway to reaction mechanism.
(ii) Catalyst raises the activation energy.
(iii) Catalyst lowers the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
Why does the rate of a reaction increase with rise in temperature?
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
In respect of the eqn k = \[\ce{Ae^{{-E_a}/{RT}}}\] in chemical kinetics, which one of the following statement is correct?
The activation energy in a chemical reaction is defined as ______.
The slope of Arrhenius Plot `("In" "k" "v"//"s" 1/"T")` of first-order reaction is −5 × 103 K. The value of Ea of the reaction is. Choose the correct option for your answer. [Given R = 8.314 JK−1mol−1]
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
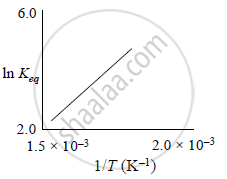
The reaction must be:
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
