Advertisements
Advertisements
प्रश्न
Why does the rate of any reaction generally decreases during the course of the reaction?
उत्तर
The rate of a reaction depends on the concentration of reactants. As the reaction progresses, the concentration of reactants decreases because the reactants start getting converted to products. Hence the rate decreases.
APPEARS IN
संबंधित प्रश्न
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
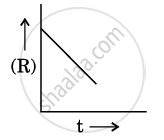
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume :
SO2Cl2 (g) → SO2 (g) + Cl2 (g)
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
Calculate the rate constant.
(Given : log 4 = 0.6021, log 2 = 0.3010)
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
For the reaction: \[\ce{2A + B → A2B}\] the rate = k[A][B]2 with k = 2.0 × 10−6 mol−2 L2 s−1. Calculate the initial rate of the reaction when [A] = 0.1 mol L−1, [B] = 0.2 mol L−1. Calculate the rate of reaction after [A] is reduced to 0.06 mol L−1.
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is doubled?
A reaction is first order in A and second order in B. How is the rate affected on increasing the concentration of B three times?
The role of a catalyst is to change
For a reaction 1/2 A ⇒ 2B, rate of disappearance of A is related 't o the appearance of B by the expression:
Which of the following statement is true?
