Advertisements
Advertisements
प्रश्न
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.
उत्तर
a. A is an alkene.
B is an aldehyde with –CH3 group.
C is a methyl ketone.
b. \[\ce{CH3CHO + [Ag(NH3)2]^{+} + OH^{-} -> CH3COO^{-} + Ag + NH3 + H2O}\]
c. \[\ce{CH3COCH3 + NaOH + I2 -> CHl3 + CH3COONa}\]
d. A: CH(CH3) = C(CH3)2, B: CH3CHO, C: O = C(CH3)2
\[\begin{array}{cc}
\ce{CH3COCH3 + CH3CHO}\phantom{............................................................................}\\
\ce{↓ Ba(OH)2}\phantom{.....................................................................}\\
\phantom{}\ce{(CH3)2C(OH)CH2COCH3 + CH3CH(OH)CH2CHO + (CH3)2C(OH)CH2CHO + CH3CH(OH)CH2COCH3}\\
\ce{↓ Heat}\phantom{.........................................................................}\\
\ce{(CH3)2C = CHCOCH3 + CH3CH = CHCHO + (CH3)2C = CHCHO + CH3CH = CHCOCH3}\phantom{............}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Give a simple chemical test to distinguish between the following pair of compounds:
Ethanal and Propanal
Predict the products of the following reactions :
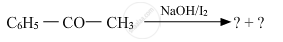
Give a simple chemical test to distinguish between the following pair of compounds :
CH3CH2CHO and CH3CH2COCH3
Propanal and Propanone
Give a simple chemical test to distinguish between the following pair of compounds:
Benzoic acid and Ethyl benzoate
Alkenes decolourise bromine water in presence of CCl4 due to formation of ______.
Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
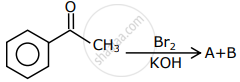
The major products formed in the following reaction sequence A and B are:
In Tollen's test for aldehyde, the overall number of electrons(s) transferred to the Tollen's reagent formula \[\ce{[Ag(NH3)2]+}\] per aldehyde group to form silver mirror is ______. (Round off to the nearest integer)
