Advertisements
Advertisements
प्रश्न
A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
पर्याय
True
False
उत्तर
This statement is False.
Explanation:
Because a neutron is an independent sub-atomic particle.
APPEARS IN
संबंधित प्रश्न
The mass of an electron is about `1/2000` times that of proton.
Fill in the blank of the following statement :
The subatomic particle not present in a hydrogen atom is ________
What is a neutron? State its relative mass and charge.
Sulphur has an atomic number of 16 and a mass number of 32. State the number of protons and neutrons present in the nucleus of sulphur.
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
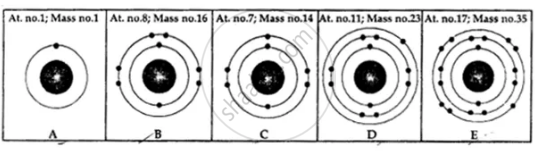
Calculate the number of neutrons, protons and electrons:
- atomic number 3 and mass number 7
- atomic number 92 and mass number 238.
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
Calculate the number of neutrons present in the nucleus of an element X which is represented as `""_15^31"X"`
James Chadwick discovered the fundamental particle called proton.
Beryllium + α – rays → Carbon + ______.
