Advertisements
Advertisements
प्रश्न
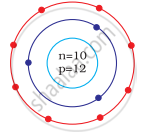
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
पर्याय
उत्तर

Explanation -
In magnesium atom `""_12^24"Mg"`
Number of n = 12, p = 12, e = 12
Number of electrons in Mg2+ = 10
APPEARS IN
संबंधित प्रश्न
Compare the properties of electrons, protons and neutrons.
The mass of an electron is about `1/2000` times that of proton.
Which subatomic particle is not present in an ordinary hydrogen atom?
Name the subatomic particle whose relative charge is : 0
What is a neutron? State its relative mass and charge.
Explain in brief the experimental proof which led to the discovery of –
Neutrons
The number of neutrons present in 73Li is ______.
Calculate the number of neutrons, protons and electrons:
- atomic number 3 and mass number 7
- atomic number 92 and mass number 238.
On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
Isotopes exist because atoms of the same element can have different numbers of ______.