Advertisements
Advertisements
प्रश्न
The mass of an electron is about `1/2000` times that of proton.
पर्याय
True
False
उत्तर
This statement is True.
Explanation:
Because it is a fact known from experiments.
APPEARS IN
संबंधित प्रश्न
Name the three sub-atomic particles of an atom.
A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
Name the subatomic particle whose relative charge is : 0
Sulphur has an atomic number of 16 and a mass number of 32. State the number of protons and neutrons present in the nucleus of sulphur.
Write down the number of neutrons in the nucleus of an atom having atomic number 17 and mass number 37.
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
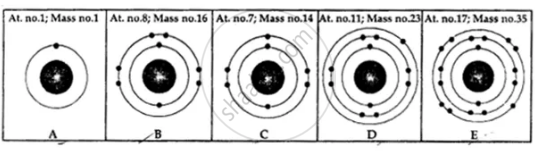
On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
Write the properties of neutrons.
Which subatomic particles have no charge?
The ratio of neutrons in C and Si with respective atomic masses 12 and 28 is ______.
