Advertisements
Advertisements
प्रश्न
Name the three sub-atomic particles of an atom.
उत्तर
Name the three subatomic particles of an atom:
- Electron
- Proton
- Neutron
APPEARS IN
संबंधित प्रश्न
The mass of an electron is about `1/2000` times that of proton.
Which subatomic particle was discovered by : Chadwick ?
Fill in the blank of the following statement :
If the nucleus of an atom has atomic number 17, mass number 37 and there are 17 electrons outside the nucleus, the number of neutrons in it is __________.
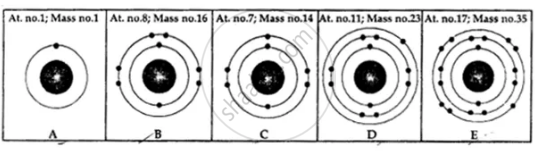
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
The number of neutrons present in 73Li is ______.
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
The hydrogen atom does not have ______.
α, β, γ rays are during the ratio active decay ______ of an atom.
Beryllium + α – rays → Carbon + ______.
The ratio of neutrons in C and Si with respective atomic masses 12 and 28 is ______.
