Advertisements
Advertisements
प्रश्न
Name the three sub-atomic particles of an atom.
उत्तर
Name the three subatomic particles of an atom:
- Electron
- Proton
- Neutron
APPEARS IN
संबंधित प्रश्न
Compare the properties of electrons, protons and neutrons.
A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
What is a neutron? State its relative mass and charge.
Sulphur has an atomic number of 16 and a mass number of 32. State the number of protons and neutrons present in the nucleus of sulphur.
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
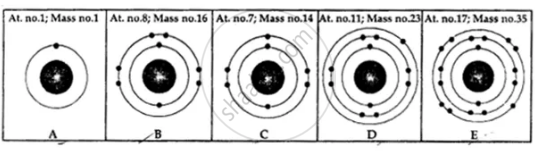
Calculate the number of neutrons, protons and electrons:
- atomic number 3 and mass number 7
- atomic number 92 and mass number 238.
Isotopes exist because atoms of the same element can have different numbers of ______.
The hydrogen atom does not have ______.
Which subatomic particles have no charge?
The stability of the nucleus is determined by the Neutron – Proton ratio.
