Advertisements
Advertisements
प्रश्न
Compare the properties of electrons, protons and neutrons.
अंतर स्पष्ट करें
उत्तर
| Property | Electron | Proton | Neutron | |
| i. | Symbol | e- or `""_1^0e` | p+ or `""_1^1p` | n or `""_0^1n` |
| ii. | Charge | -1 unit or - 1.6 × 10-19 C | +1 unit or + 1.6 × 10-19 C | Zero |
| iii. | Mass | 9.1 × 10-31 kg | 1.67 × 10-27 kg | 1.675 × 10-27 kg |
| iv. | Location | present around the nucleus | present in nucleus | present in nucleus |
| v. | Discovery | J.J. Thomson | E. Goldstein | Chadwick |
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Name the three sub-atomic particles of an atom.
Which subatomic particle is not present in an ordinary hydrogen atom?
Explain in brief the experimental proof which led to the discovery of –
Neutrons
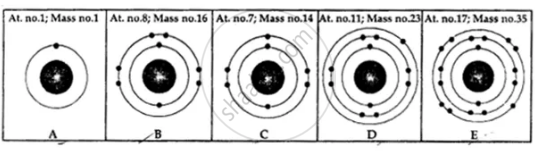
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.
Calculate the number of neutrons, protons and electrons:
- atomic number 3 and mass number 7
- atomic number 92 and mass number 238.
Write the properties of neutrons.
Which subatomic particles have no charge?
α, β, γ rays are during the ratio active decay ______ of an atom.
The stability of the nucleus is determined by the Neutron – Proton ratio.
The ratio of neutrons in C and Si with respective atomic masses 12 and 28 is ______.
